I have posted a new paper about thermochemistry, electrochemistry and thermodynamics in star evolution.
http://vixra.org/pdf/1408.0157v1.pdf
I have also noticed a simple mistake in which I note decomposition reactions as being (synthesis) reactions, in which it is not. A decomposition reaction is analysis, and a combination reaction is synthesis.
Decomposition reaction:
Combination (synthesis) reaction:
The vast majority of chemical reactions in star evolution are synthesis reactions which are exothermic (heat releasing).
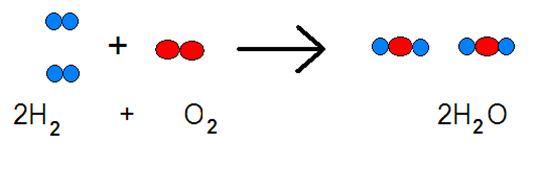
A very good example is one I found:
While three bonds must be broken (two H―H and one O-O double bond), a total of four bonds are made (four O―H bonds). Since all the bonds are similar in strength, making more bonds than are broken means the release of energy. In mathematical terms
ΔH° = 2DH―H + DO=O + 4DO―H
= 2 × 436 kJ mol–1 + 1 × 498 kJ mol–1 – 4 × 467 kJ mol–1
= – 498 kJ mol–1
In summary, there are two factors which determine whether a gaseous reaction will be exothermic or not:
(1) the relative strengths of the bonds as measured by the bond enthalpies, and
(2) the relative number of bonds broken and formed. An exothermic reaction corresponds to the formation of more bonds, stronger bonds, or both.
When water bonds are formed they release energy.
http://chempaths.chemeddl.org/services/chempaths/?q=book/General%20Chemistry%20Textbook/Thermodynamics%3A%20Atoms%2C%20Molecules%20and%20Energy/1491/bond-enthalpies-and


No comments:
Post a Comment
Helpful comments will be appreciated, but if the user does not want to address the issues being presented they will be ignored. This is a blog dedicated to trying to explain how to make sense of the discovery that planet formation is star evolution itself, not a blog for false mainstream beliefs.